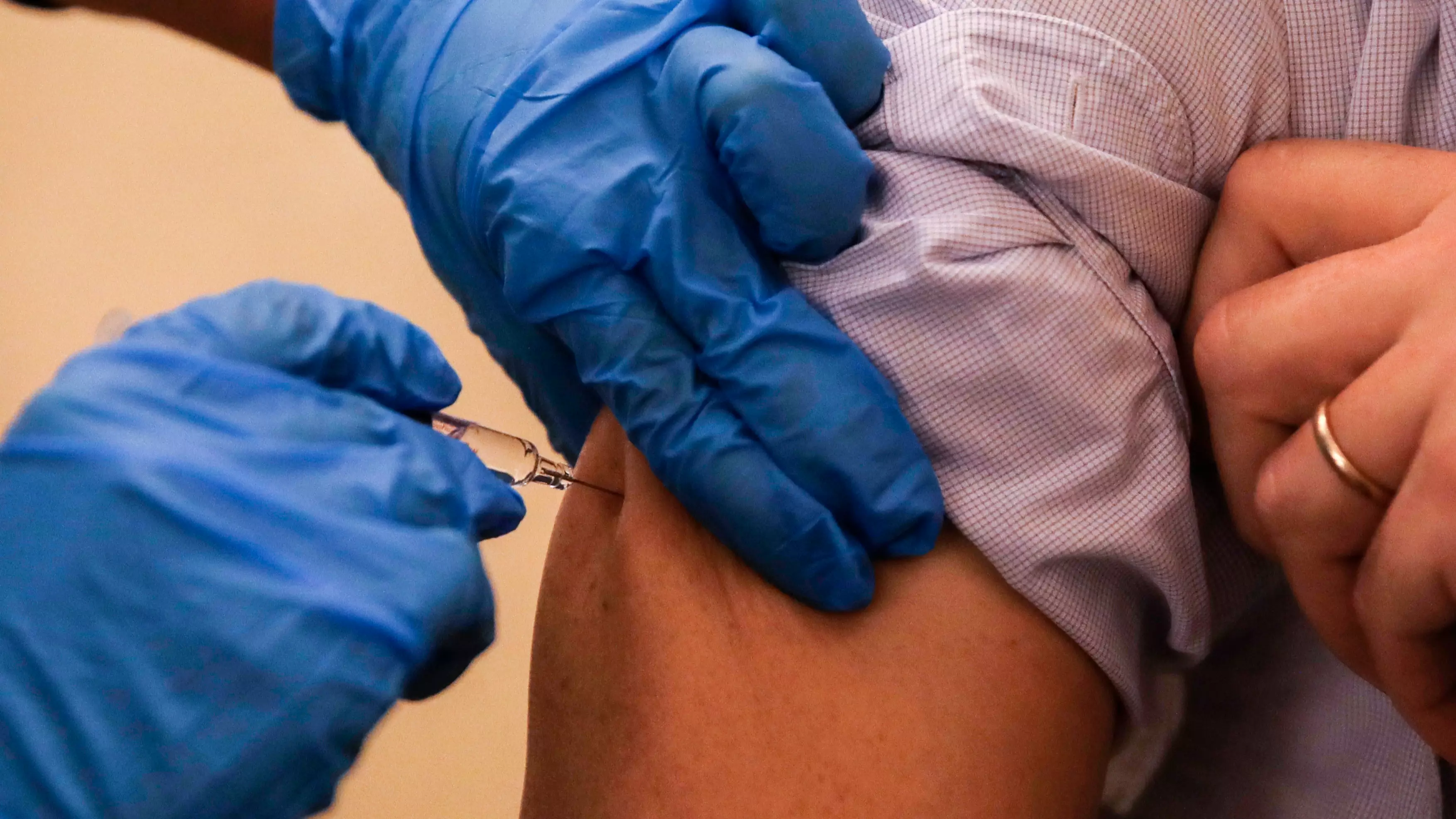
The first Covid-19 vaccine could be available this year and data from clinical trials suggests it will protect 90 percent of people from the deadly disease.
The vaccine has been developed by Pfizer and BioNTech and the UK has already ordered 30 million doses.
According to the firms, the jab has been tested out on 43,500 people in six countries, with no safety concerns detected - and only 94 people in the trial went on to develop Covid-19.
Advert
The companies are now planning on applying for emergency approval to roll the vaccine out by the end of the month.
If successful, Pfizer is confident it will be able to manufacture 50 million doses by the end of the year and a further 1.3 billion by the end of 2021.
While it is one of many being tested by a number of firms, this one uses an experimental approach by injecting part of the disease's genetic code into an individual in order to train the body's immune system.
Advert
According to the results from the initial analysis, two doses, three weeks apart, are needed for it to be effective.
And trials in the US, Germany, Turkey, South Africa, Brazil and Argentina, have shown that 90 percent effectiveness is achieved a week after the second jab.
There is, however, no definitive answer on how long the immunity lasts.
With the vaccine having to be stored at below minus 80C, there are also questions about how it will be delivered.
Advert
Speaking about the breakthrough, Dr Albert Bourla, the chairman of Pfizer, said he hoped this would help bring about an end to the suffering being experienced around the world.
He said: "Today is a great day for science and humanity. The first set of results from our Phase 3 Covid-19 vaccine trial provides the initial evidence of our vaccine's ability to prevent Covid-19.
"With today's news, we are a significant step closer to providing people around the world with a much-needed breakthrough to help bring an end to this global health crisis.
Advert
"While we are all in the midst of a second wave and many of us in lockdown, we appreciate even more how important this milestone is on our path towards ending this pandemic and for all of us to regain a sense of normality."
Last month, Johnson & Johnson's coronavirus vaccine trial was temporarily put on hold after one of the participants developed an 'unexplained illness'.
This was the second time a vaccine trial, which is in the late stages of testing, has been affected by such a setback.
A top official, however, said this is 'not at all unusual' when dealing with so many people, and the trial has since been resumed.
Advert
The company has enrolled 60,000 people to see whether its vaccine would prove effective against the pandemic.
Featured Image Credit: PATopics: Science, Coronavirus, Politics, Health