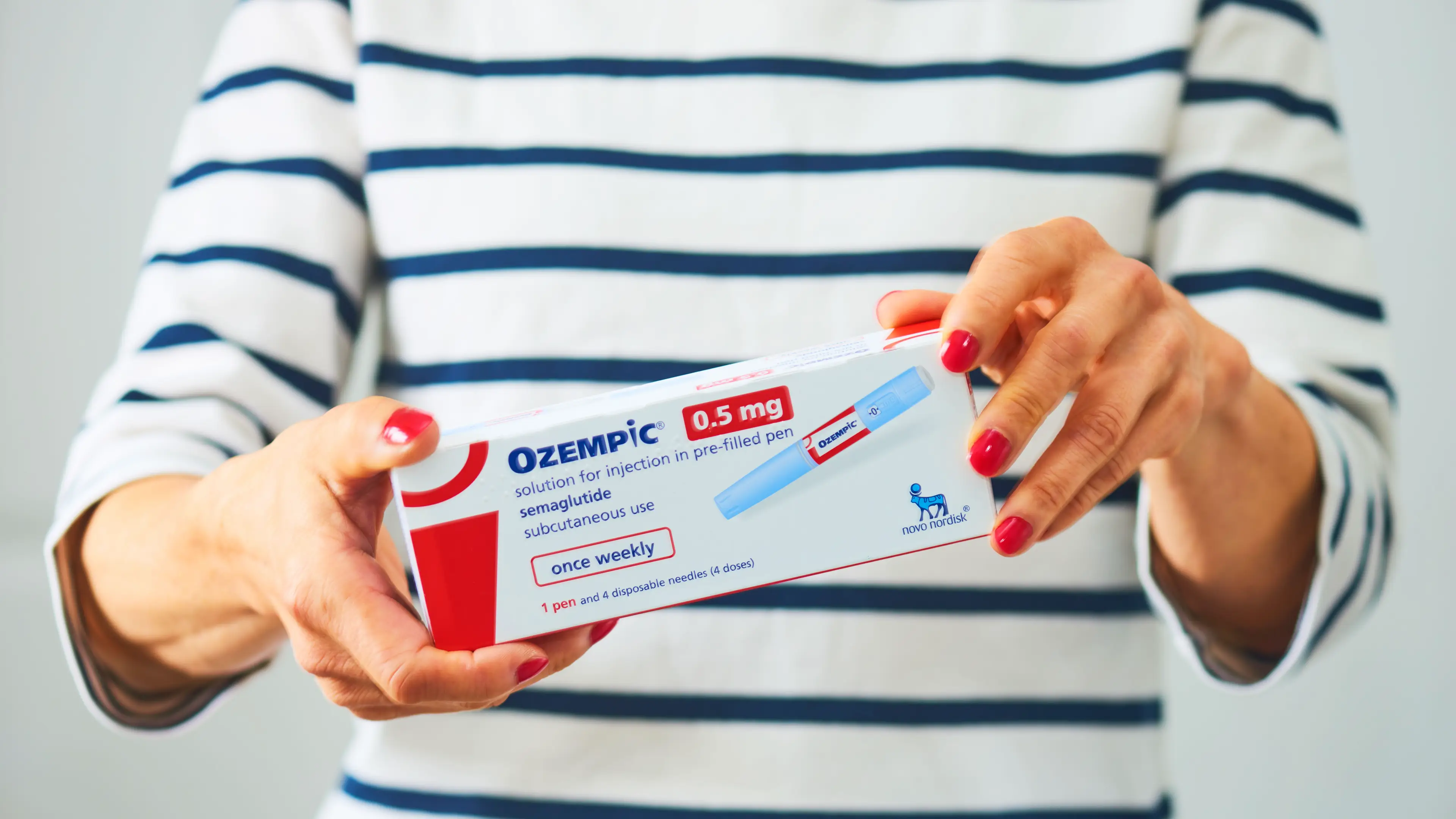
The manufacturers of Ozempic and Mounjaro are planning to launch a new weight loss method in 2026.
While Ozempic remains a GLP-1 medicine for adults with Type-2 diabetes, and not a weight loss drug, Mounjaro - the brand name for tirzepatide - can be prescribed for weight-loss via the NHS.
These drugs typically come in jab form, but it appears the new era of medicine could be upon us fairly soon.
Mounjaro's manufacturer, Eli Lilly, announced in September that it was hoping to 'offer a convenient, once-daily pill that can be scaled globally'.
Advert
Named 'Orforglipron', it can be 'taken once per day without food and water restrictions'.
While it remains in development, initial tests showed that it 'lowered weight by an average of 10.5 per cent (22.9 lbs) compared to 2.2 per cent (5.1 lbs) with a placebo', Eli Lilly explained.
Kenneth Custer, executive vice president at Eli Lilly, added: "With these positive data in hand, we are moving with urgency toward global regulatory submissions to potentially meet the needs of patients who are waiting."
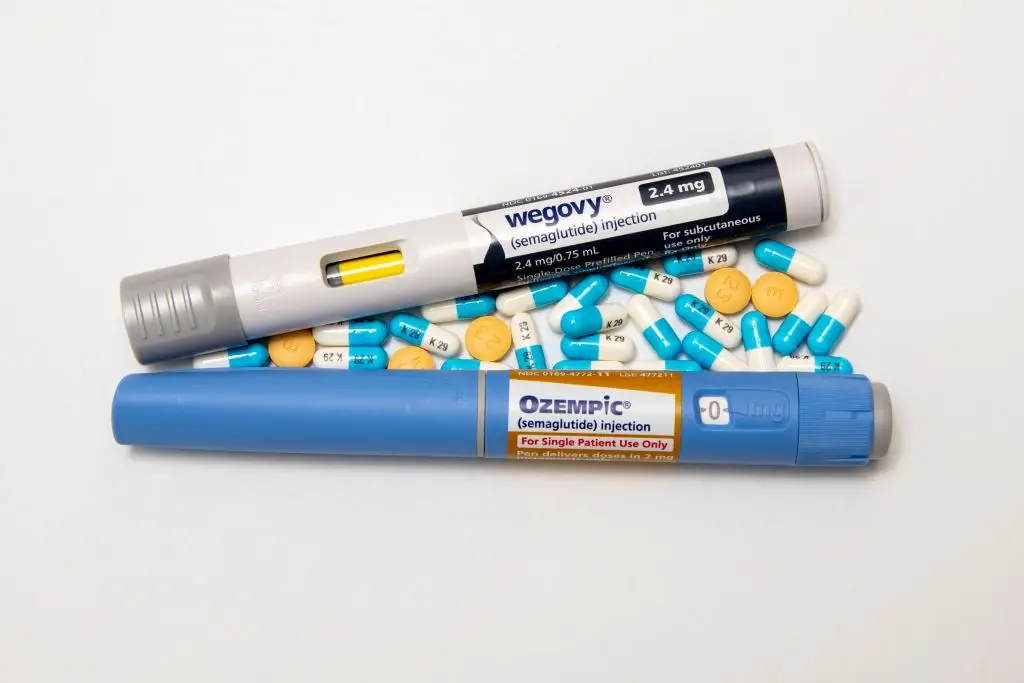
It comes after Eli Lilly said that its experimental pill, orforglipron, outperformed Novo Nordisk’s oral semaglutide in a year-long trial.
At the highest dose, orforglipron delivered greater weight loss - an average 9.2 percent (about 19.7lbs) compared with Ozempic's 5.3 percent (about 11 lbs).

When including all participants, Lilly’s pill produced 8.2 percent weight loss versus 5.3 percent for semaglutide.
“For the majority of patients, this could be the main medicine that they need to control their Type 2 diabetes as well as their obesity,” Eli Lilly Chief Scientific Officer Dan Skovronsky
Ozempic.
Drugmakers had hoped the oral version of the drug may have had a new use in the form of slowing down the progression of Alzheimer’s - however, it was reported that an oral version failed to do so in two major trials.
The Danish drugmaker said patients taking the pill saw no meaningful reduction in disease advancement, which put a hold on expectations that the bestselling diabetes medication might have a new use.
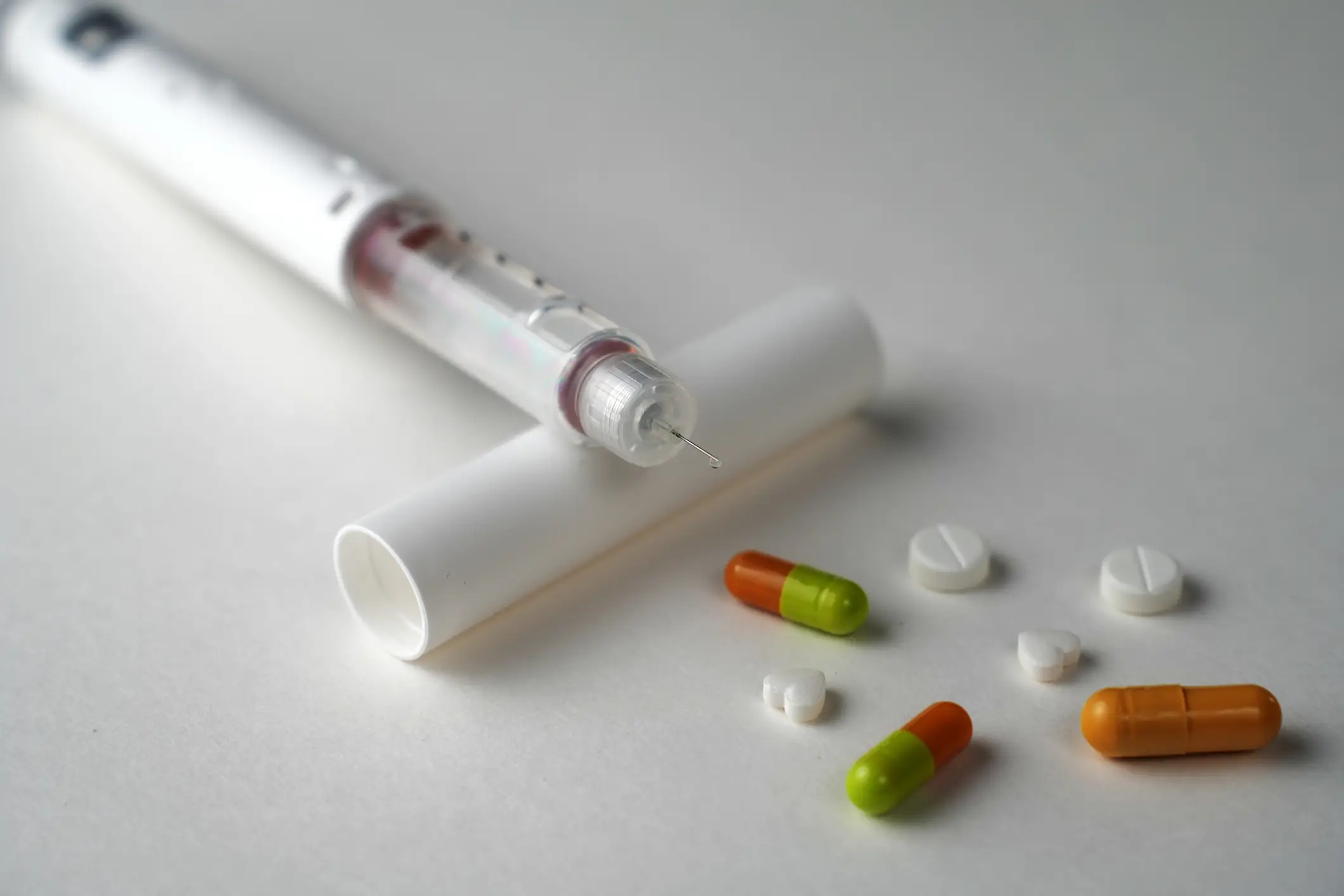
As many companies are rushing to bring an anti-obesity pill to market, it seems there would be demand for it, particularly for people who don't like needles.
“With this newer generation of medications, we’re not just focusing on weight loss,” David Lau, an endocrinologist and professor emeritus at the University of Calgary Cumming School of Medicine told The Washington Post.
“We’re talking about changes beyond what you see on the scale.”
Despite all the optimism, the drugs are still subject to FDA approval.
LADbible Group has contacted Novo Nordisk and Eli Lilly for comment.