News
The latest national and international news stories that will inform you, make you laugh and make you think.
The latest national and international news stories that will inform you, make you laugh and make you think.
The latest national and international news stories that will inform you, make you laugh and make you think.

Shirilla's behaviour behind bars has been revealed in a new report

He took the club to its 20th league title in his debut season
breaking
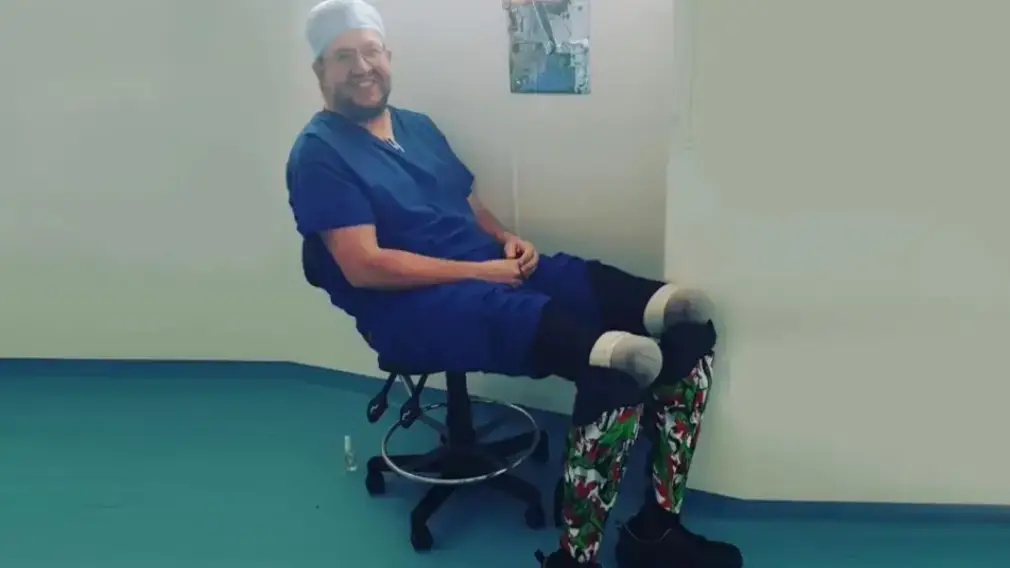
Neil Hopper was a surgeon who amputated his own legs and has now been struck off

Shocking video showed the moment Shirilla's parents confronted police officers at the station