
A woman was diagnosed with numerous brain tumours after using injectable birth control for two decades.
Last year Kerry Sharples made an appointment with the her GP after experiencing throbbing pain behind her left ear. She later began to experience a similar pulsing sensation in her right ear months later but ignored it for months after initial tests came back clear.
However when the mum-of-two mentioned the sensation at a check-up in August 2025 she was referred for blood tests and an MRI scan – which revealed she had not one but four brain tumours, called meningioma.
"I was in complete shock," Kerry said of the diagnosis. "I was like 'what does this mean'. I still don't know if I've completely dealt with it.
Advert
"I'm very much a crack on and get on with it but sometimes it does get to you."

What are meningioma tumours?
According to John Hopkins Medicine, meningioma are tumours which grow in the brain's connective tissues (meninges). They are the most common type of primary brain tumour, accounting for approximately 30 percent of all cases.
Symptoms vary depending on the location, however these are the most common:
- Headaches
- Seizures
- Blurred vision
- Weakness in your arms or legs
- Numbness
Most meningioma tumours, including Kerry's, are benign (non-cancerous), according to Cancer Research, with women being more commonly affected than men.
Recent studies into medroxyprogesterone contraceptive injections, a progestogen-only form of contraception which is injected every 12–13 weeks, revealed that women who've used the drug for more than three years are at an increased risk of developing a meningioma. However this considered to be a rare side effect.
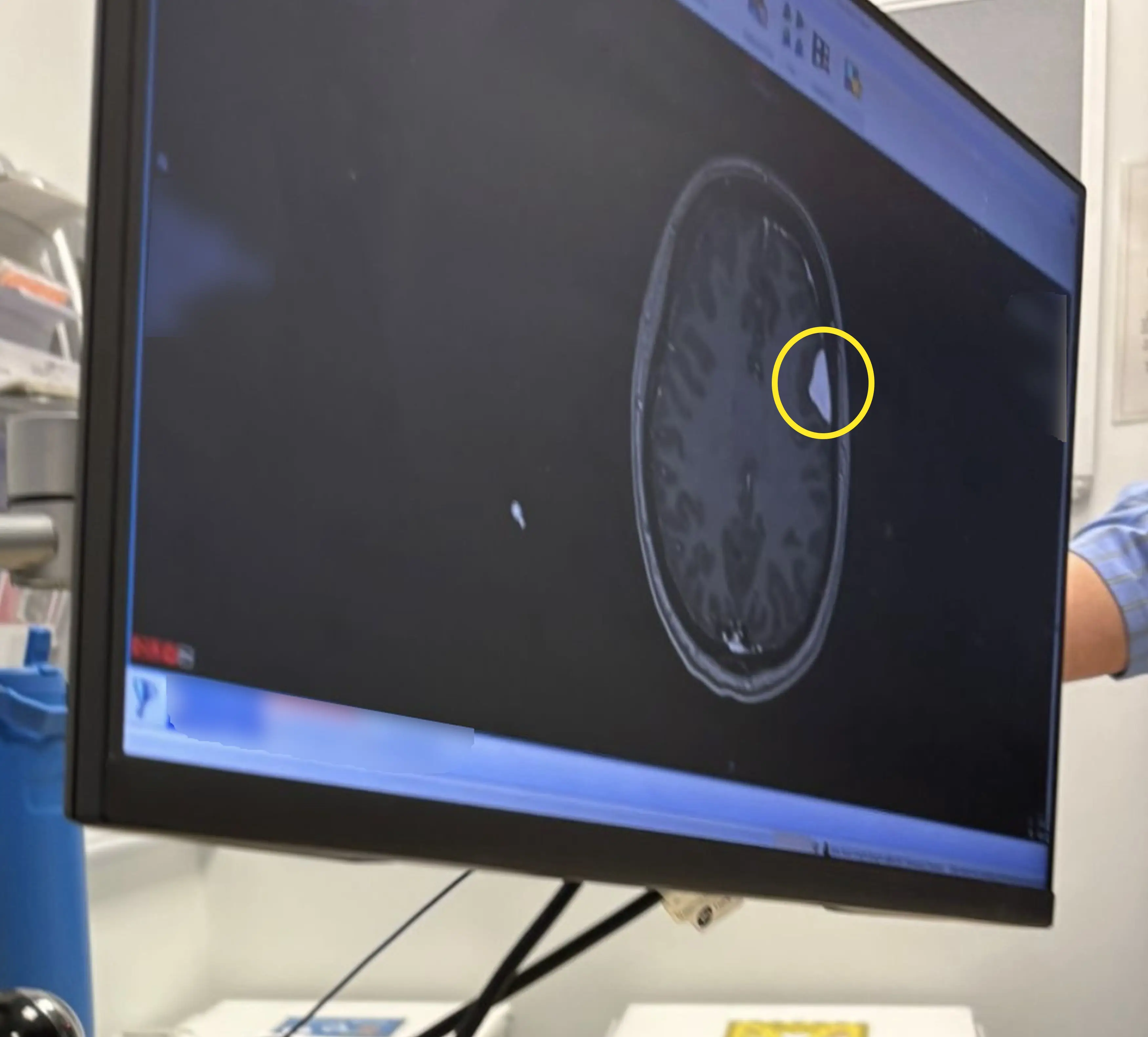
For 45-year-old Kerry, who'd been taking the shots for 21 years as she didn't want to have periods, said that she now regrets ever taking the birth control.
"I regret taking these injections. It sounds so silly now that I just didn't want the periods and stayed on it and now I've got four brain tumours," she said.
"I'd take the periods any day now. It's crazy."
She stopped taking the injections in December 2025 and now wants to raise awareness to other women about the potential risks.
"I don't want to scare people [or] to put the fear of God into them but they do need to be aware of what they can cause," she said of anyone taking injectable birth control.
"Just re-think and do your research. There are other options out there. I'm just going to stay off everything because I'm just wary of everything. You just don't know."

Kerry also questioned why she hadn't been warned about the potential risks of injectable birth control causing brain tumours earlier, noting that in 2024 one manufacturer acknowledged a small increased risk of developing meningioma after prolonged medroxyprogesterone use.
"It's really unfair. I know there's side effects to everything. But I just feel like it's very much like 'you've got that and just deal with it and get on with it'," she added.
"I've been on it for over 20 years. Why was that not brought to my attention even in 2024? Why has it not been told to me?"
In response to Kerry's case, the Medicines and Healthcare products Regulatory Agency (MHRA) gave the following statement: "Patient safety is our top priority. MHRA continuously monitors all licensed medicines for safety.
"[The drug] is an injectable contraceptive injection which contains medroxyprogesterone acetate (MPA) and is used to prevent unwanted pregnancy.
"Product information for products containing MPA was initially updated in 2024 following studies which showed a risk of meningioma associated with use of high dose MPA.

"It was further updated in early 2025 with new advice and additional measures to manage the risk.
"The Patient Information Leaflet was also updated to ensure patients were aware of the symptoms of meningioma and with the advice that they should contact their doctor if they experienced these symptoms.
"For the majority of people, the benefits of [the drug] far outweigh the risk, but we advise anyone with any concerns to speak with their GP, pharmacist or contraceptive provider."
"Patient safety is our top priority," added a spokesperson for Pfizer. "We conduct rigorous and continuous monitoring of all our medicines, including assessments of reported adverse events, in collaboration with health authorities around the globe.
"Medroxyprogesterone acetate has been approved in more than 60 countries over the last 30 years, has a well-established efficacy and safety profile and has been a treatment option for millions of patients during that time.
"People should talk to their doctor, pharmacist or nurse if they have any concerns or experience any side effects."
Topics: Health, Sex and Relationships